|
Hence, by this concept molecular weight is the force acting on a given chemical substance having a molecular mass. The weight of any given substance is the force (typically gravitational force) acting on the given substance having a specific mass. Also, the difference between molecular weight and molecular mass is a matter of simple definition of weight. Therefore, for ionic substances, the molecular weight cannot be obtained and the formula weight is generally taken as the molecular weight. They dissociate as soon as they are dissolved into a solution. This is because the ionic substances do not have chemical bonds and do not exist as individual molecules. The weight usually obtained from the chemical formula of ionic substances is the formula weight. But in the case of ionic substances, the formula weight is different from the molecular weight. And generally the weight obtained from the chemical formula or molecular formula is both the formula weight and the molecular weight of the substance. As given above for you how to calculate molecular weight, it is clear that it can be done using the molecular formula of the chemical substance. 
One point of note is that, there are minor differences between molecular weight, formula weight and molecular mass. Thus, molecular weight helps in determining the amount of a chemical compound required for a given chemical reaction. The mole in turn is used to determine the stoichiometry of a given molecule in chemical reactions and equations. The molecular weight in turn helps you in knowing how many grams of a chemical substance is present in one mole of a compound. This isotope has an assigned value of 12 amu and is thus regarded as standard. Both the atomic weight and the molecular weight are defined with respect to the mass of isotope of carbon - 12. Here, amu is the atomic mass unit and Da is Dalton. Typically, the molecular weight formula gives the value in units of amu or Da. Thus, in simple terms, the molecular weight formula is nothing but the sum of the total number of atoms present in a given molecule which can be known from the given molecular formula. The number of each type of atom present in a molecule is multiplied by the atomic weight and is then added to the weights of other atoms. Thus, the molecular weight definition and formula can both be derived from the molecular formula of a given molecule. This information can be easily obtained from the molecular formula. The molecular weight of a given molecule is the sum of the atomic weights of all the atoms present in a given molecule. How to Calculate Molecular Weight Formula? Similarly, the molecular weight of Fe i.e. For example, in the case of O 2, the molecular weight is 32, which is the sum of the atomic weight of a single Oxygen atom. In case of single elements, the molecular weight is the sum of the atomic weights of the total number of atoms of an element required to form a stable molecule. Thus, the molecular weight meaning clearly states that it is the total of all the atomic weights of each of the atoms present in a given molecule of a chemical compound. In the following table for this reaction:Ĭlick on image below to see a pictorial representation of this.The molecular weight definition can be given as the sum of the atomic weight values of the atoms in a given chemical compound. If all elements are balanced, the equation is balanced. If each side of the equation has the same number of atoms of a given element, that element is balanced. Step 2: Count the number of atoms of each type on each side of the equation (for the reactants and for the products). In the total number of atoms present for each type of molecule.Ĭlick here if you would like to see molecules broken into atoms. Count the number of each type of atom in each type of molecule. Step 1: Break each molecule up into the individual atoms. 
To see if the equation is balanced, we can follow two steps: If this equation is balanced, the same atoms (in number and identity) will be present as reactants and products. Was the chemical equation previously discussed balanced? Let's take a closer look: That is written so this is true is said to be balanced. Need to be present at the end of the reaction as products. All of theĪtoms that were present at the beginning of the reaction as reactants also 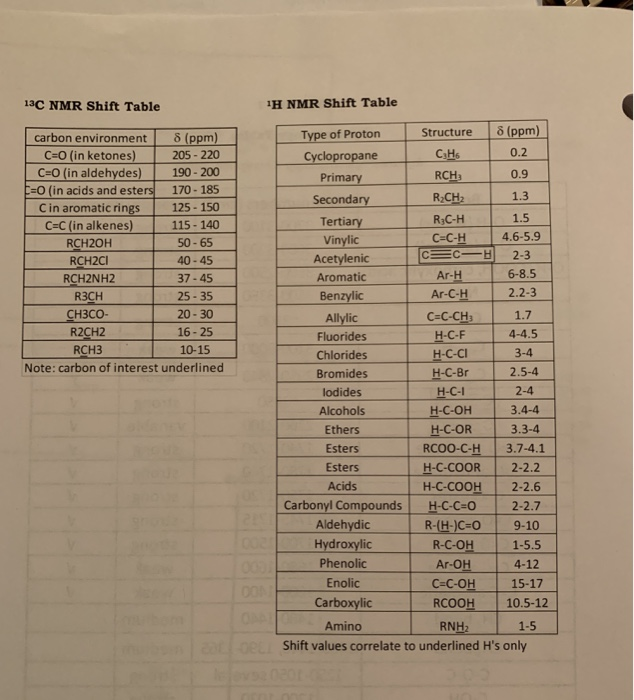
This law has an effect on the coefficients of a chemical equation. In a chemical reaction, atoms can notīe created or destroyed they simply rearrange themselves to form new products. You surely have heard that matter can not be created or destroyed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
